Pfizer plans to make all its patented medicines available at a not-for-profit price for the world’s poorest nations. That will include treatments for infectious diseases and cancers
A person walks past a Pfizer logo amid the coronavirus disease pandemic in the Manhattan borough of New York City, New York, U.S., April 1, 2021. REUTERS/Carlo Allegriwill make all of its patented medicines including COVID-19 treatment Paxlovid and big-selling breast cancer drug Ibrance available at a not-for-profit price to 45 of the world's poorest countries, the drugmaker said on Wednesday.
Pfizer said its plan includes 23 wholly-owned, patented medicines and vaccines that treat infectious diseases, certain cancers, and rare and inflammatory diseases. In addition to Paxlovid and Ibrance, the list includes pneumonia vaccine Prevnar 13, rheumatoid arthritis drug Xeljanz and cancer treatments Xalkori and Inlyta.Chief Executive Albert Bourla said in an interview that all the medicines being made available should be of use.
When Pfizer launches new medicines and vaccines, they will also be included in the drug portfolio at a not-for-profit price, it said. Malawi President Lazarus Chakwera said in a statement the accord will allow the countries and the drugmaker to share "the burden of costs and tasks in the production and delivery of supplies that will save millions of lives."
France Dernières Nouvelles, France Actualités
Similar News:Vous pouvez également lire des articles d'actualité similaires à celui-ci que nous avons collectés auprès d'autres sources d'information.
 Pfizer: 3 COVID vaccine doses are 80 percent effective for kids 5 and underThree doses of the Pfizer-BioNTech COVID-19 vaccine were roughly 80 percent effective at preventing infections for children ages 6 months to 5 years old, according to trial data Pfizer released Monday.
Pfizer: 3 COVID vaccine doses are 80 percent effective for kids 5 and underThree doses of the Pfizer-BioNTech COVID-19 vaccine were roughly 80 percent effective at preventing infections for children ages 6 months to 5 years old, according to trial data Pfizer released Monday.
Lire la suite »
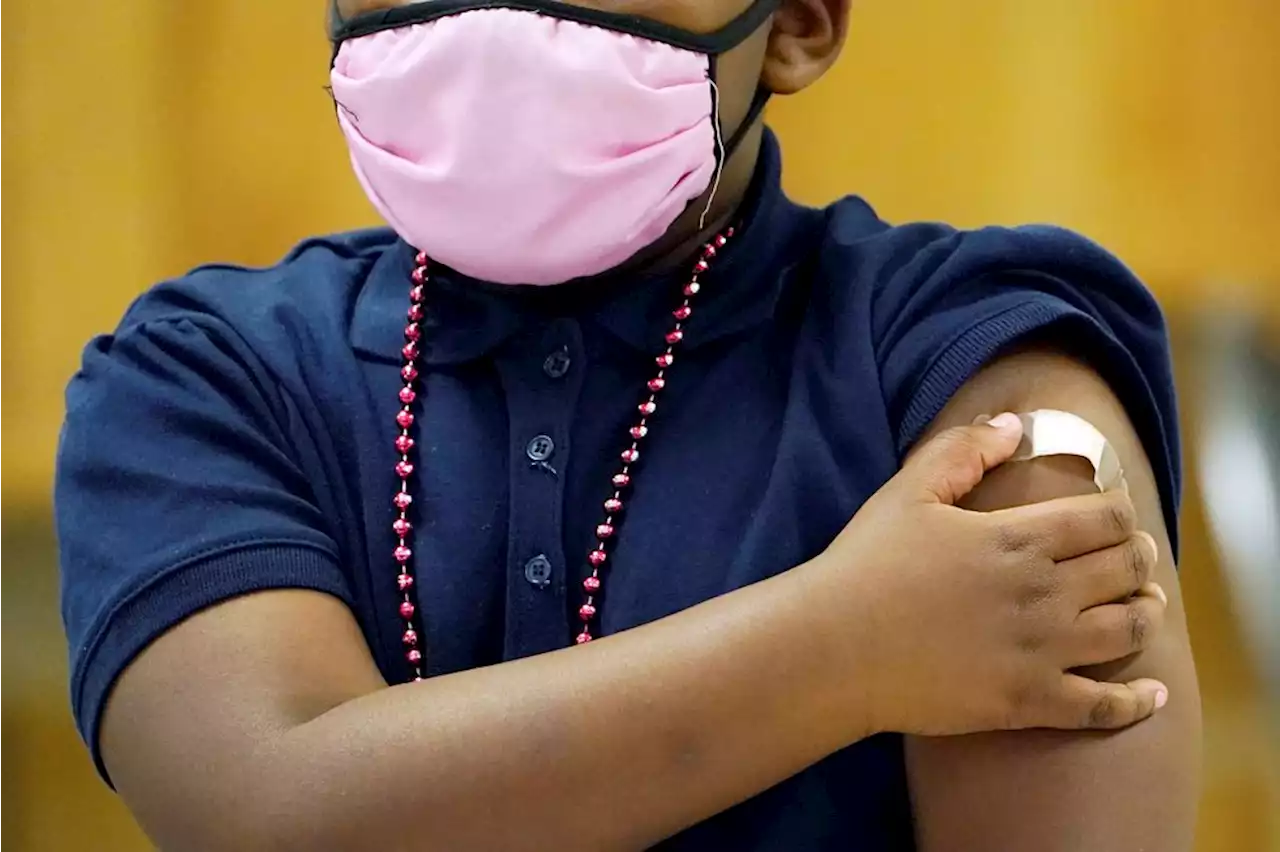 Pfizer says 3 COVID shots protect children under 5Pfizer says three small doses of its COVID-19 vaccine protect kids under 5 and that it plans to give the data to U.S. regulators later this week.
Pfizer says 3 COVID shots protect children under 5Pfizer says three small doses of its COVID-19 vaccine protect kids under 5 and that it plans to give the data to U.S. regulators later this week.
Lire la suite »
 Pfizer/BioNTech say 3 COVID shots elicit good response in children under 5Drugmakers Pfizer and BioNTech said on Monday that three doses of their COVID-19 vaccine generated a strong immune response in children under age 5 and was safe and well-tolerated in their clinical trial.
Pfizer/BioNTech say 3 COVID shots elicit good response in children under 5Drugmakers Pfizer and BioNTech said on Monday that three doses of their COVID-19 vaccine generated a strong immune response in children under age 5 and was safe and well-tolerated in their clinical trial.
Lire la suite »
 Pfizer says 3 COVID shots protect children under 5Three doses of Pfizer’s COVID-19 vaccine offer strong protection for children younger than 5, the company said Monday, another step toward shots for the littlest kids possibly beginning in early summer.
Pfizer says 3 COVID shots protect children under 5Three doses of Pfizer’s COVID-19 vaccine offer strong protection for children younger than 5, the company said Monday, another step toward shots for the littlest kids possibly beginning in early summer.
Lire la suite »
 Pfizer: 3 COVID shots protect children under 5Pfizer says three small doses of its COVID-19 vaccine protect kids under 5. The company released preliminary results on Monday and said it plans to give the data to U.S. regulators later this week. It's the latest step toward letting the littlest kids get the shots. The 18 million tots under 5 are the only group in the U.S. not yet eligible for COVID-19 vaccination. The Food and Drug Administration has begun evaluating data from rival Moderna. That company hopes to offer two kid-sized shots by summer. The FDA has set tentative dates next month for its scientific advisers to publicly debate data from Pfizer and Moderna.
Pfizer: 3 COVID shots protect children under 5Pfizer says three small doses of its COVID-19 vaccine protect kids under 5. The company released preliminary results on Monday and said it plans to give the data to U.S. regulators later this week. It's the latest step toward letting the littlest kids get the shots. The 18 million tots under 5 are the only group in the U.S. not yet eligible for COVID-19 vaccination. The Food and Drug Administration has begun evaluating data from rival Moderna. That company hopes to offer two kid-sized shots by summer. The FDA has set tentative dates next month for its scientific advisers to publicly debate data from Pfizer and Moderna.
Lire la suite »
 Pfizer to Sell Vaccines, Drugs at Low Prices to Poorer CountriesPfizer will sell nearly two dozen of its patent-protected drugs and vaccines at not-for-profit prices to some of the world’s poorest countries
Pfizer to Sell Vaccines, Drugs at Low Prices to Poorer CountriesPfizer will sell nearly two dozen of its patent-protected drugs and vaccines at not-for-profit prices to some of the world’s poorest countries
Lire la suite »