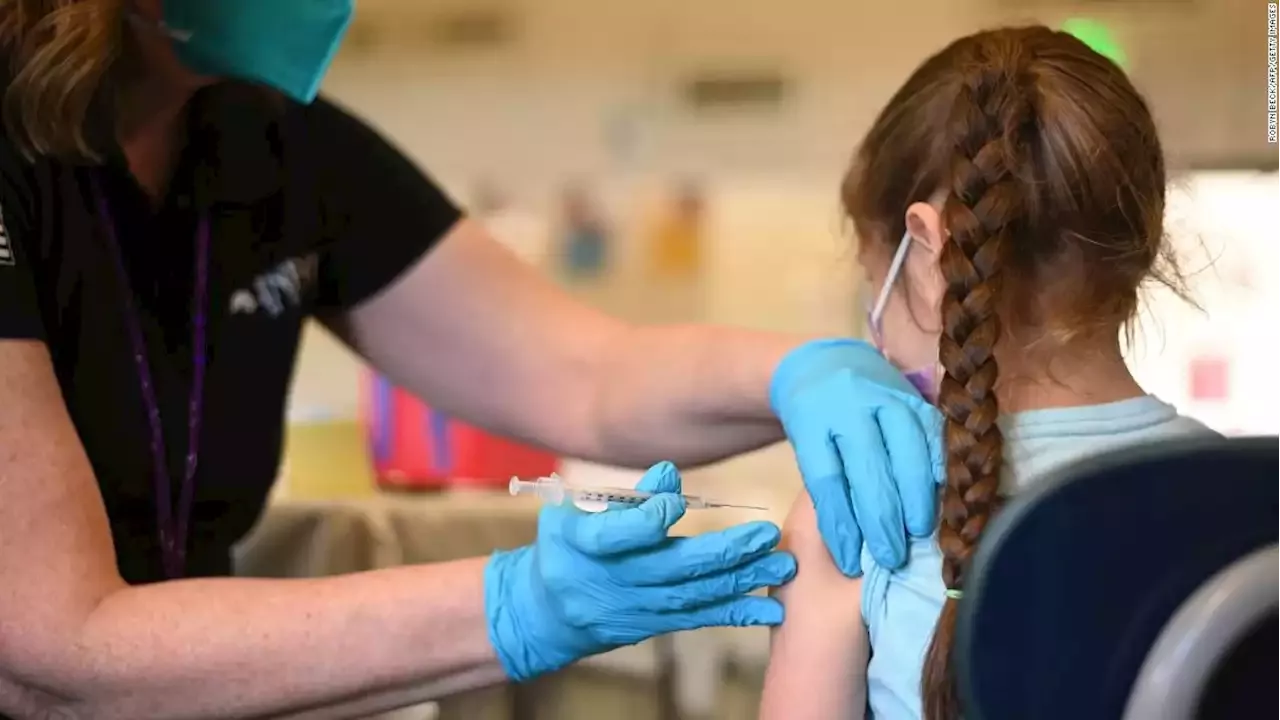
The US Food and Drug Administration has granted emergency use authorization for a booster dose of Pfizer/BioNTech's Covid-19 vaccine for children ages 5 to 11
Pfizer requested this EUA at the end of April, citing company data that showed that a third vaccine dose raised Omicron-fighting antibodies by 36 times in this age group."While it has largely been the case that COVID-19 tends to be less severe in children than adults, the omicron wave has seen more kids getting sick with the disease and being hospitalized, and children may also experience longer term effects, even following initially mild disease," FDA Commissioner Dr.
Moderna has also requested FDA authorization for a second Covid-19 booster shot for everyone 18 and older, but a decision has yet to be made regarding that request.
France Dernières Nouvelles, France Actualités
Similar News:Vous pouvez également lire des articles d'actualité similaires à celui-ci que nous avons collectés auprès d'autres sources d'information.
 FDA authorizes Pfizer COVID-19 booster shots for children ages 5 to 11The FDA has granted emergency use authorization for a booster dose of Pfizer/BioNTech's COVID-19 vaccine for children ages 5 to 11 at least five months after completion of the primary vaccine series.
FDA authorizes Pfizer COVID-19 booster shots for children ages 5 to 11The FDA has granted emergency use authorization for a booster dose of Pfizer/BioNTech's COVID-19 vaccine for children ages 5 to 11 at least five months after completion of the primary vaccine series.
Lire la suite »
 FDA approves Pfizer COVID-19 booster for children ages 5 to 11Pfizer’s COVID-19 booster shot for U.S. kids ages 5 to 11 has been approved by the FDA. Next, it will need the CDC’s final sign-off.
FDA approves Pfizer COVID-19 booster for children ages 5 to 11Pfizer’s COVID-19 booster shot for U.S. kids ages 5 to 11 has been approved by the FDA. Next, it will need the CDC’s final sign-off.
Lire la suite »
 FDA authorizes COVID-19 booster for children ages 5 to 11The Food and Drug Administration (FDA) amended Pfizer's COVID-19 booster shot emergency use authorization to allow for children ages 5 to 11 to receive the vaccine.
FDA authorizes COVID-19 booster for children ages 5 to 11The Food and Drug Administration (FDA) amended Pfizer's COVID-19 booster shot emergency use authorization to allow for children ages 5 to 11 to receive the vaccine.
Lire la suite »
 FDA authorizes Pfizer COVID-19 booster shots for children ages 5 to 11The FDA has granted emergency use authorization for a booster dose of Pfizer/BioNTech's COVID-19 vaccine for children ages 5 to 11 at least five months after completion of the primary vaccine series.
FDA authorizes Pfizer COVID-19 booster shots for children ages 5 to 11The FDA has granted emergency use authorization for a booster dose of Pfizer/BioNTech's COVID-19 vaccine for children ages 5 to 11 at least five months after completion of the primary vaccine series.
Lire la suite »
 Steve Kerr returns to Warriors after testing positive for COVID-19 last weekWarriors coach Steve Kerr, who has cleared the NBA’s health and safety protocols, described his symptoms as “mild.”
Steve Kerr returns to Warriors after testing positive for COVID-19 last weekWarriors coach Steve Kerr, who has cleared the NBA’s health and safety protocols, described his symptoms as “mild.”
Lire la suite »
 North Korea Reports More COVID-19 DeathsNorth Korea reported 15 more COVID-19 deaths on Sunday, bringing the country’s death toll to 42. State media also reported another 296,180 infections, adding up to a total of 820,620 in recent weeks. After claiming no coronavirus cases for the entire pandemic, North Korea announced its first COVID-19 patients on Thursday.
North Korea Reports More COVID-19 DeathsNorth Korea reported 15 more COVID-19 deaths on Sunday, bringing the country’s death toll to 42. State media also reported another 296,180 infections, adding up to a total of 820,620 in recent weeks. After claiming no coronavirus cases for the entire pandemic, North Korea announced its first COVID-19 patients on Thursday.
Lire la suite »